Frequently Asked Questions
Question
Answer
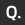
- What is "nickel-metal hydride battery?"

- A nickel-metal hydride battery consists of electrodes, separators, and electrolyte. Hydrogen storage alloy is used on the negative electrode, and nickel hydroxide is used on the positive electrode. The separator is of polyolefin non-woven material, and the electrolyte is an alkaline solution, mostly KOH. Unlike lead-acid batteries and nickel-cadmium batteries, no environmentally harmful materials are used. It is capable of rapid charging and discharging of large currents, and has a long life cycle. With its recent application on hybrid vehicles, nickel-metal hydride batteries are considered an important component in today’s industrial world.
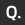
- How does the chemical reaction occur?

-
Discharge Electrochemical Reaction
During discharge, hydrogen moves from the negative electrode (MH) to the positive electrode (NiOOH). The positive electrode becomes Ni(OH)2.
Charge Electrochemical Reaction
During charge, hydrogen moves from the positive electrode (Ni(OH)2) to the negative electrode (M). The negative electrode becomes MH.
Electrochemical reaction is simpler than those of lead-acid batteries, which uses dissolution and precipitation processes of heavy metal. In principle, the electrochemical reaction is everlasting as long as the electrodes do not change.
- What is special about the GIGACELL, compared with other rechargeable batteries?

-
GIGACELL can charge and discharge at high rates. It does not apply hazardous materials, and because the electrolyte is water-based, there is no risk of fire.

Type GIGACELL Li-ion Lead-acid
(long life cycle type)NAS Strong Points High-capacity, rapid charge / discharge Rapid charge / discharge High reliability High energy density Active Material MH、Ni(OH)2 Li Pb Na、S Electrolyte KOH Organic solvent H2SO4 Beta alumina Rated Voltage (V) 1.2 3.6 2 2 Operating Temperature (degrees Celsius) Room temp. Room temp. 0-40 280-350 Self-discharge Yes Yes Yes No Discharging Rates Continuous: approx. 3 C
Instantaneous: approx. 20 CContinuous: approx. 3 C
Instantaneous: approx. 20 CContinuous: approx. 0.2 C
Instantaneous: approx. 6 CContinuous: approx. 0.1 C
Instantaneous: approx. 0.4 C
- How long is GIGACELL’s life cycle?

- A battery’s life cycle will depend on various factors such as charge / discharge frequency and battery temperature. Please contact us on your exact application.
- What is memory effect?

- Memory effect is an effect that occurs when charges and discharges are repeated within a shallow range, without using the whole battery capacity. As a result of this, battery voltage will drop at those shallow ranges, regardless of the remaining capacity. Nickel-metal hydride batteries are said to have this effect.
When voltage drops caused by this effect occur, operation can be limited for equipments set at high voltage.
Test results indicate that GIGACELL’s voltage drop is very small, and by reflecting it to the minimum voltage settings of equipments, it is possible to prevent problems that occur from the memory effect.
Also, by regularly charging the GIGACELL (full discharge and full charge), it is possible to eliminate the memory effect.
- Is it safe?

- There are monitoring devices for each module that measures the voltage and current of the battery. By knowing the state of the battery at all times, over-charging / discharging can be prevented.
GIGACELL is a trademark of Kawasaki Heavy Industries, Ltd. in Japan and the U.S.A.







